Background
In 2009, a novel influenza A (H1N1) virus subtype emerged to become the first global pandemic to be declared in over four decades (Charania and Tsuji 2011). The World Health Organization (WHO) first reported the novel influenza strain on March 18th, 2009 and declared the global outbreak of influenza A (H1N1) a pandemic on June 11th, 2009 (Standards 2010). By the end of that year, the pandemic had caused over 16,000 deaths worldwide (Wynn and Moore 2012). In Canada, the 2009 H1N1 influenza pandemic (pH1N1) caused 8,678 hospitalizations, 1,473 (17.0%) intensive care unit admissions, and 428 (4.9%) deaths (Scott 2010). Responding to this pandemic cost Canada an estimated $2 billion (Health and King 2010, Standards 2010, Wynn and Moore 2012). Prior to official pandemic status, on May 1st 2009, Canada had reported 51 confirmed cases of pH1N1 (Standards 2010). Canada’s first peak occurred in June 2009, primarily in Manitoba, and the second from mid-October to mid-November (Embree 2010).
A study of the characteristics of Canadian patients critically ill with pH1N1 found that young, female, and Aboriginal patients without significant comorbidities (i.e., relatively healthy) were the populations most affected (Kumar, Zarychanski et al. 2009). A systematic review of clinical and epidemiological factors of pH1N1 also reported a high infection rate in children and young adults, with fewer elderly people infected (Khandaker, Dierig et al. 2011).
Canada’s public health response to pH1N1, recommended by the Public Health Agency of Canada (PHAC), was prevention through vaccination, conducting the highest mass immunization campaign in its history, with 40–45% of the population vaccinated for pH1N1 (Scott 2010). Health Canada approved the adjuvanted pH1N1 vaccine on October 22nd, 2009 and by October 29th, 2009, the first long lines for vaccines had formed (Standards 2010). In Canada, and globally, vaccination of health care workers (HCWs) was prioritized as a key strategy of the pandemic H1N1 (pH1N1) preparedness process (CDC 2009, Spika and Butler-Jones 2009, Zarocostas 2009, Kendal and MacDonald 2010, Scott 2010, Bryce, Embree et al. 2012). The importance of vaccinating HCWs for influenza control is widely acknowledged (Saxen and Virtanen 1999, Orr 2000, Hofmann, Ferracin et al. 2006, Poland, Ofstead et al. 2008, Poland 2010, Lam, Chambers et al. 2010a). In addition to self-protection, vaccination of HCWs is important to protect patients as seemingly healthy HCWs may still act as vectors and/or reservoirs for the influenza virus (Weinstein, Bridges et al. 2003, Lam, Chambers et al. 2010a). Furthermore, the influenza vaccine is known to be most effective in healthy adults (such as HCWs), with lower efficaciousness in the elderly and immune-compromised (Nichol and Treanor 2006, Monto 2010). Influenza vaccination of HCWs is also cost-effective, with one economic evaluation using UK data finding a worst case scenario cost of £405/life-year gained (Burls, Jordan et al. 2006). Despite these benefits, influenza vaccination rates of HCWs remain suboptimal (Burls, Jordan et al. 2006, Thomas, Jefferson et al. 2010, Lam, Chambers et al. 2010a), prompting some to advocate for mandatory influenza vaccination of all HCWs (Bryce, Embree et al. 2012), while others argue that influenza vaccination of HCWs should not be coerced (Gallant, Vollman et al. 2008, Yassi, Lockhart et al. 2010).
This paper reviews all relevant qualitative research that looks at Canadian healthcare workers’ lived experiences during the 2009 H1N1 pandemic with respect to HCWs’ uptake of the pandemic vaccine. This is the first review of the qualitative research on this subject undertaken by the National Collaborating Centre for Infectious Diseases (NCCID) to date.
Methods
Studies that used qualitative research methods to investigate the experiences of Canadian healthcare workers with respect to pandemic vaccination during the 2009 – 2010 influenza “A” (H1N1) pandemic were included in this review. Relevant studies were identified using the following medical subject headlines (MeSH) and key words: “H1N1,” “pandemic,” “influenza A,” “healthcare worker,” “health care worker,” “public health,” “public health nurs*,” “experience,” “Canada,” “Canad*,” “vacc*,” “qualitative,” and “(interview* or focus group*)”. The search time frame was limited from January 2009 to present. Databases searched were those available through New York University (NYU) School of Medicine and BOBST library, including Ovid, Medline, PubMed, Embase, EBSCO (CINAHL), Scopus, Web of Science, Web of Knowledge, JStor, and ProQuest Dissertations and Theses. In addition, relevant articles were searched for by topic through NYU’s BOBST library under the following subjects: sociology, anthropology, nursing/medicine, social work, and public health. Finally, additional literature was sought using Google Scholar and by scanning bibliographies of included studies and relevant reviews (Bish, Yardley et al. 2011, Blasi, Aliberti et al. 2012, Brien, Kwong et al. 2012, Music 2012, Prematunge, Corace et al. 2012, Herzog, Alvarez-Pasquin et al. 2013, Hollmeyer, Hayden et al. 2013).
Once identified, studies were reviewed to ensure that the inclusion criteria were met, more specifically that data collection used qualitative research methods and the focus was specific to the 2009 influenza A (H1N1) pandemic in Canada.
Results
Only one study was found that met the inclusion criteria for this review, a thesis by Prematunge (2013) that used qualitative methods as one part of a three-part study to investigate healthcare workers’ attitudes and barriers to influenza vaccination. This thesis acted as partial requirements for a Masters of Science degree in Epidemiology at the University of Ottawa and as a component of an independent study, Optimizing H1N1 Vaccination Uptake among Healthcare Workers (referred to from here on as HCW-pH1N1 Study), for which the author was the research coordinator. Two components of this study have been published, a systematic review of the factors influencing HCW vaccination uptake or refusal in the current scientific literature (Prematunge, Corace et al. 2012) and quantitative analyses to identify factors that predict HCW vaccination (Corace, Prematunge et al. 2013).
Study Background, Design, and Methodology Strategy
The HCW-pH1N1 Study was conducted at the Ottawa Hospital (TOH), a bilingual tertiary-care centre in Ottawa, Ontario. TOH launched a mass immunization campaign targeting all hospital staff, as outlined by Canada’s national pH1N1 response plan, in October 2009. This campaign worked to enhance staff immunization through distinct strategies, including highly visible communication tactics, weekend immunization clinics accommodating staff family vaccinations, extended vaccination clinic hours, and roving vaccine carts, all of which was supported by hospital management and administration leadership (Prematunge 2013). The HCW-pH1N1 Study, led by McCarthy, Garber, and Corace, investigated vaccination uptake by HCWs at TOH during the 2009/2010-influenza vaccination campaign to identify factors contributing to acceptance of the pH1N1 and 2008/2009 seasonal influenza vaccine.
The Ottawa Hospital Research Ethics Board approved all aspects of the study. Bilingual questionnaire packages were mailed to all active employees (i.e., multi-professional HCWs) in July 2010 and 3,382 of 10,464 surveys (32.4% response rate) were returned by the July 30th, 2010 deadline to the hospital’s Occupational Health and Safety Departments (OCHS). The OCHS compared the surveys to the 2009/2010 staff influenza status records to verify the respondents’ self-reported vaccination status and then de-identified the questionnaires. Qualitative data were collected via open-ended questions included in the questionnaire.
The questionnaire, collaboratively prepared based on a literature review and the expert opinions of the interdisciplinary team, contained the following measures:
- socio-demographics, including age, gender, race, occupation, marital status, TOH campus/site of employment, and perceived health status;
- self-reported seasonal flu vaccine history, including influenza vaccination history, allergies to vaccine components, and previous immunization experiences;
- the pH1N1 Vaccine Attitudes and Knowledge Scale, a 34 item scale addressing behaviors and attitudes determining influenza vaccine uptake which required participants to rank their agreement to each scale item using a 5-point Likert scale;
- the Seasonal Influenza (sINFLU) Vaccine Attitudes and Knowledge Scale, a 34 item scale similar to (3) but addressing their agreement to statements regarding sINFLU vaccination;
- reasons for vaccine uptake, an open-ended qualitative question asking HCW participants to report and rank their top three reasons for receiving the pH1N1 and/or sINFLU vaccines;
- reasons for vaccine avoidance, an open-ended qualitative question asking HCW participants to report and rank their top three reasons for avoiding the pH1N1 and/or sINFLU vaccines; and
- a pH1N1 client satisfaction questionnaire, a 9-item scale based on the validated Client Satisfaction Questionnaire (CSQ) and modified to more accurately reflect pH1N1-specific heath care services.
The pH1N1 Vaccine Attitudes and Knowledge and the Seasonal Influenza (sINFLU) Vaccine Attitudes and Knowledge Scales were adapted from existing questionnaires investigating influenza vaccine uptake among HCWs (Mok, Yeung et al. 2006, Looijmans-van den Akker, van Delden et al. 2009). Only sections (5) and (6) collected qualitative data, thus they are the only sections relevant to this review.
The objective of Prematunge’s thesis (2013), consistent with the HCW-pH1N1 study and using the qualitative data collected, was to increase HCWs’ uptake of influenza vaccinations via informing effective design and implementation of vaccination campaigns. Nosocomial influenza outbreaks are not addressed. This thesis addresses the following primary research questions: 1) why do HCWs receive or not receive influenza vaccination during influenza pandemics, specifically pH1N1; and 2) why do HCWs receive or not receive sINFLU vaccination during non-pandemic influenza seasons? In addition, Prematunge (2013) states the following objectives for her thesis: 1) identify the range of factors motivating HCWs to receive and/ or not receive pH1N1 vaccination, as available in the current literature; 2) capture key themes and rationales reflecting HCW decisions towards pH1N1 and sINFLU vaccination and non-vaccination; and 3) explain the relationship(s) between key barriers and motivators towards pH1N1 and sINFLU vaccination among HCWs in pandemic and non-pandemic situations.
As most current research on HCW pH1N1 vaccination uses quantitative analysis, the use of qualitative research included in this thesis permits broader discussion of Canadian HCWs’ worldviews, values, perceptions, beliefs, rationales, and assumptions about vaccines, further contributing to the understanding of HCWs’ views on influenza vaccination (Padgett 2012). Participant HCWs were asked, using free-form writing, to identify and rank their top three reasons for either uptake or avoidance of both the pH1N1 vaccine and the 2008/2009 sINFLU vaccine. With these responses, content analysis was performed. Content analysis has been defined as a “research technique for making replicable and valid inferences from data to their contexts” (Krippendorff 2012). The specific steps, or analytical constructs, to conducting content analysis can be based on existing theories, expert knowledge/ experience, or previous research, and developed into a priori schema for categorizing and quantifying data. Thus, these analytical constructs allow data (such as quantified text) to be converted to the research question answers (Marsh and White 2006).
Data Analysis
Prematunge (2013) used content analysis methodology as described by Neuendorf to code the qualitative reasons provided by the participant HCWs, with each HCW-provided reason considered a single “meaning unit” that could be quantified (Neuendorf 2002). Consistent with content analysis methodology, a priori coding was used and several existing systematic reviews of HCW influenza vaccination behavior informed the development of the preliminary coding scheme (Brewer, Chapman et al. 2007, Hollmeyer, Hayden et al. 2009, Thomas, Jefferson et al. 2010, Lam, Chambers et al. 2010a, Prematunge, Corace et al. 2012). Revisions of the preliminary coding scheme were done with a random subsample of “meaning units” (n=100) and consultation with influenza vaccination research and qualitative research methodology specialists. A random subsample of units (n=500) was then independently co-coded using the revised coding schemas, which were further refined and finalized by consensus. Inter-coder reliability was reported with Cohen’s Kappa (κ) calculations. Finally, the primary coder (Permutange) coded all of the “meaning units” using the finalized sets of a priori coding schemas including: 1) pH1N1 vaccination motivators, 2) pH1N1 vaccination barriers, 3) sINFLU vaccination motivators, and 4) sINFLU vaccination barriers. When motivators or barriers inconsistent with the a priori schema were identified, these were classified as “alternative” motivators/barriers. An audit trail was maintained to ensure transparency and repeatability. All coding and analysis processes were conducted using Microsoft Office Excel (version 2003) and SPSS for Windows (version 17.0) to calculate counts and proportions of key themes to determine frequencies of reported barriers and motivators to pH1N1 and sINFLU vaccination.
Qualitative Analysis Findings
Of the 3,275 survey packages that were returned and included in the qualitative analysis, 2,862 (87.4%) HCWs were vaccinated against pH1N1, 2,433 (74.3%) received the 2008/2009sINFLU vaccine, and 1,745 (53.8%) received the 2009/2010sINFLU vaccine. The demographic statistics revealed that of the HCW respondents, mean age was 42.93 +/- 11.23, 2,608 (81.4%) were female, 2,884 (89.3%) were Caucasian, 2,516 (76.2%) reported having formal religious beliefs, 2,454 (75.9%) considered themselves to be in a relationship, 1,556 (48.8%) had dependents less than 21 years of age, and 2,335 (71.7%) worked full-time. Of the HCW study participants, 1,153 (35.2%) were nurses, 172 (5.3%) were physicians, 361 (11.0%) were allied HCWs, 721 (22.0%) held administrative/clerical positions, 241 (7.4%) were healthcare technicians, 276 (8.4%) worked in research and/or laboratories, 216 (6.6%) worked in facilities and/or logistics, and 135 (4.1%) reported other non-clinical roles.
After final coding, a total of 19,531 “meaning units” were coded into pH1N1 and sINFLU vaccination motivators (reasons cited by vaccinated HCWs on why they accepted vaccination) or barriers (reasons cited by unvaccinated HCWs on why they refused vaccination). For pH1N1 vaccination specifically, 8,424 “meaning units” from vaccinated HCWs were coded as pH1N1 vaccination motivators and 1,268 “meaning units” from unvaccinated HCW were coded as vaccination barriers. For sINFLU vaccination specifically, 7,259 “meaning units” were coded as sINFLU vaccination motivators and 2,580 “meaning units” were coded as sINFLU vaccination barriers.
Cohen’s Kappa (κ) , with 100% indicating total agreement between two researchers and 0% indicating no agreement, was calculated to be 85% for pH1N1 vaccination motivators and 73% for pH1N1 vaccine barriers. Cohen’s Kappa (κ) was calculated to be 78% for sINFLU vaccination motivators and 64% for sINFLU vaccine barriers.
Prematunge (2013) notes that many vaccination motivators and barriers, describing HCWs’ reasons for accepting or refusing vaccination respectively, were similar for both pH1N1 and sINFLU vaccines, with a few motivators or barriers being specific to either pH1N1 or sINFLU, as described in the following sections. Three figures at the end of the sections illustrate these findings. Figure 1 describes motivators and barriers to accepting pH1N1 and sINFLU vaccination, highlighting those that are shared and those that are vaccine-specific. Figure 2 further describes the specifics of each motivator. Figure 3 further describes the components of each barrier.
Motivators for Accepting pH1N1 Vaccination, Reported by Vaccinated HCWs
The following motivators were cited in conjunction with accepting pH1N1 vaccination: to protect personal health, to protect family and loved ones, to protect patients, for reasons attributed to their occupation as a HCW, to protect the community/society at large, and, specific to pH1N1, “alternative” motivators (Figure 2).
The primary motivation for accepting vaccination reported by HCWs vaccinated for pH1N1 (28.1%) was for personal and/or self-care reasons, which was also the most often first ranked motivation for pH1N1 vaccination uptake. Specifically, self-care themes represented included: personal health, personal safety and protection, infection prevention in self, and reduced infection risk to self. Furthermore, HCWs that accepted both pH1N1 and sINFLU vaccination tended to be more mindful of potential complications due to influenza when concurrent risk factors were present. Vaccine acceptance was thus more likely when HCWs also reported having one or more of the risk factors for influenza, such as chronic illness, age, or asthma. Specific to pH1N1 vaccination, the following personal motivators were mentioned: fear of infection, the pandemic nature of pH1N1, and perceptions of pH1N1 being more severe than seasonal influenza.
Secondly, motivation to protect family and loved ones was commonly cited (20.3 %) as a reason for receiving pH1N1 vaccination, and was the most frequently second ranked reason for accepting vaccination. Common reasons mentioned included safety and protection of loved ones, preventing transmission to loved ones, and acknowledged increased risk among loved ones, such as young children, elderly relatives, chronic health conditions, among others.
Thirdly, prevention of transmission to patients also motivated HCWs to accept vaccination against pH1N1 (10.8 %) and was the most often third ranked reason to become vaccinated. Reasons mentioned included patient safety and protection and recognizing their own role as HCWs in preventing influenza transmission, and potentially subsequent complications, to patients at point of work/care.
Fourthly, many HCWs (9.8%) reported being motivated to become vaccinated for pH1N1 because of their status as HCWs and other occupational factors. The work place influenced HCWs’ choices in several ways. Particularly, vaccinated HCWs reported motives related to potential increases in workload resulting from influenza-related absenteeism, professional obligation, recognized occupational hazard related to the nature of the job, and work place requirements and/or culture. Some HCWs also reported that pH1N1 immunization was compulsory or strongly suggested by their employer. Employer-mandated pH1N1 vaccination was more commonly reported as being associated with pH1N1 vaccination than sINFLU vaccination. Encouragement from colleagues and supervisors were also positive motivators for HCWs to become vaccinated.
Fifthly, protection of their community and society at large was a key motivator for many HCWs, with 7% of all ranked reasons to be vaccinated for pH1N1 specifically mentioning limiting general influenza spread in others.
Finally, an “alternative” motivator to be vaccinated during the pandemic was mentioned by HCWs: convenient access to pH1N1 vaccine, described as short line-ups, reduced wait times, ability to become vaccinated at work, and free access to the vaccine.
Barriers to Accepting pH1N1 Vaccination, Reported by Unvaccinated HCWs
Pandemic H1N1 vaccination barriers identified and discussed by HCWs who refused vaccination included personal/self and vaccine-related barriers (Figure 3).
In addition to being among the highest ranked motivators, unvaccinated HCWs also mentioned personal reasons as a barrier to pH1N1 (28.6%) vaccination. Many non-vaccinated HCWs reported their personal immune function was sufficient and could handle an influenza infection and so the vaccine was not necessary for them personally. Other personal reasons that HCWs reported declining vaccination included that: influenza vaccination was not necessary for them; they do not get the flu; they are never sick; they perceived themselves to be at low risk for infection; they “do not believe in immunization”; refusal is based on personal choice; and they had previously experienced side-effects to an influenza vaccine, such as a sore arm, headache, or feeling sick.
Vaccine-related barriers were also mentioned for pH1N1 (28.7%) vaccinations, such as vaccine safety, vaccine ingredients, possible adverse-effects, or allergies to the vaccines as barriers to accepting vaccination. In particular, HCWs mentioned concern regarding exposure to chemicals (e.g. thimerosal, mercury, and adjuvants) used in manufacturing vaccines, vaccine-induced adverse reactions experienced by others (e.g. HCW friends, family members), lack of vaccine effectiveness, and a belief of vaccination causing influenza or influenza-like illness. Vaccine- related barriers that were specific to pH1N1 vaccination primarily related to a lack of trust of the vaccine, as reflected by 14.4% of non-vaccinated HCW citing perceptions of limited knowledge of the pH1N1 vaccine, particularly with respect to knowledge gaps in vaccine manufacturing and vaccine research and development processes. Perceptions that the pH1N1 vaccine was novel and different from sINFLU, rushed into development, and not as vigorously assessed for vaccine safety issues or long-term side-effects compared to conventional sINFLU vaccines were reported by many non-vaccinated HCWs.
Also specific to pH1N1 vaccination was an alternative barrier describing a lack of clear and consistent communication about the vaccine. Miscommunication also centred around pH1N1 in that it was perceived to be exaggerated by various groups (e.g. media, pharmaceutical companies, government organizations, and other public health authorities) that may have had hidden agendas and/or stood to profit from greater uptake of vaccine.
Motivators for Accepting sINFLU Vaccination, Reported by Vaccinated HCWs
All of the motivators identified for accepting pH1N1 vaccination were also mentioned for sINFLU vaccination, including: to protect personal health, to protect family and loved ones, to protect patients, for reasons attributed to their occupation as a HCW, and to protect the community/society at large (Figure 2).
The primary motivation reported by HCWs who were vaccinated for sINFLU vaccination (28.1%) was for personal and/or self-care reasons, which was also the most frequently first ranked motivator for accepting sINFLU vaccination. Specifically, personal reasons cited included: personal health, personal safety and protection, infection prevention in self, and reduced infection risk to self. Vaccine acceptance was also more common when HCWs reported also having concurrent co-morbidities that increased the risk factor for influenza, such as chronic illness, age, or asthma.
Motivation to protect family and loved ones was the second most commonly cited reason for receiving sINFLU (17.6%) vaccination, and was the most frequently second ranked reason for accepting vaccination. Common reasons mentioned included safety and protection of loved ones, preventing transmission to loved ones, and acknowledged increased risk among loved ones, such as young children, elderly relatives, chronic health conditions, among others.
Ranked third, many HCWs (11%) reported being motivated to become vaccinated for pH1N1 by their HCW status and other occupational factors. Particularly, vaccinated HCWs reported motives related to potential increases in workload resulting from influenza related absenteeism, professional obligation, recognized occupational hazard related to the nature of the job, and work place requirements and/ or culture. Some HCWs reported that influenza immunization was compulsory or strongly suggested by their employer, although less than for pH1N1. A pro-vaccination workplace culture, such as encouragement from colleagues and supervisors, was also a positive motivator for HCWs to become vaccinated.
Fourthly, prevention of transmission to patients also motivated HCWs to accept vaccination against sINFLU (10.7 %). Reasons mentioned included patient safety and protection and recognizing their own role as HCWs in preventing influenza transmission, and potentially subsequent complications, to patients at point of work/care.
Lastly, protection of their community and society at large was a key motivator for many HCWs, with 6.4% of all ranked reasons for accepting sINFLU vaccination specifically mentioning limiting general influenza spread in others.
Barriers to Accepting sINFLU vaccination, reported by Unvaccinated HCWs
Barriers to accepting sINFLU vaccination identified by unvaccinated HCW included personal/self, vaccine-related, and alternative barriers (Figure 3). The primary barrier to sINFLU vaccination reported was personal reasons, with 28.4% of unvaccinated HCWs citing this. Many non-vaccinated HCWs reported their personal immune function was sufficient and could handle an influenza infection and so the vaccine was not necessary for them personally. Other personal reasons that HCWs reported declining vaccination included that: influenza vaccination was not necessary for them; they do not get the flu; they are never sick; they perceived themselves to be at low risk for infection; they “do not believe in immunization”; refusal is based on personal choice; and they had previously experienced side-effects to an influenza vaccine, such as a sore arm, headache, or feeling sick. Notably, a sINFLU vaccination-specific personal barrier to getting vaccinated was to promote one’s “natural immunity,” with some HCWs reporting that they do not receive the sINFLU vaccine to enhance their immune function, which they assumed would occur through exposure to the virus without concurrent immunization.
Vaccine-related barriers were mentioned for accepting sINFLU vaccinations by 20% of non-vaccinated HCWs. Many HCWs mentioned vaccine safety, vaccine ingredients, possible adverse-effects or allergies to the vaccines as barriers in both pandemic and non-pandemic scenarios. Specific vaccine-related barriers mentioned by HCWs included concern regarding exposure to chemicals (e.g. thimerosal, mercury, and adjuvants) used in manufacturing vaccines, vaccine-induced adverse reactions experienced by others (e.g. HCW friends, family members), lack of vaccine effectiveness, and a belief that vaccination causes influenza or influenza-like illness.
Specific to sINFLU vaccine, its perceived inability to prevent influenza was cited as a barrier based on knowledge that sINFLU vaccines are designed to vaccinate against the viral strains predicted to be prevalent in the upcoming flu season, largely based on viral strains that were most prevalent during the previous influenza season. Non-vaccinated HCWs reported little faith in the prediction process.
Lastly, an alternative barrier specific to sINFLU identified by unvaccinated HCWs is that they simply do not receive sINFLU vaccination out of habit.
Study Limitations
Prematunge (2013) identifies several potential limitations to the study, including potential volunteer bias due to the possibility of more vaccinated HCWs choosing to participate than non-vaccinated HCWs. In addition, social acceptability may have influenced HCW participants’ responses, thus inaccurately reflecting their true motives and introducing error into the findings. Prematunge (2013) also notes that the nature of qualitative research, as described by Neuendorf (2002), means that personal and social contexts of the research may have influenced the development of the a priori coding schema and the content analysis processes. Moreover, since the coding development process was largely based on prior, quantitative studies, the analysis may not be sufficiently sensitive to more subtle findings. Prematunge acknowledges the possible existence of alternative themes that were not identified or fully explored by their study, although she notes the study findings are consistent with existing quantitative and qualitative literature on HCW pH1N1 and sINFLU vaccination behavior (Prematunge 2013). Although Prematunge (2013) also cites the recruitment of study participants from a single geographic area and a subsequent lack of generalizability of the findings as a limitation, qualitative research methods are particularly valued for their depth of findings and, unlike quantitative studies, generalizability is not a concept applied in qualitative research methods (Charmaz 2006, Padgett 2012).
In addition to the above, it is worth noting that these qualitative data were collected without direct person-to-person contact and the inability to observe participant body language, tone, and reactions, may have potentially further limited its sensitivity (Padgett 2012). Furthermore, in addition to social acceptability influencing the responses, it was impossible for the researchers to verify that the same HCW participant who answered the rest of the survey and whose vaccination status was linked to the questionnaire answered the qualitative questions themselves. Other questions and scale items on the questionnaire may have also influenced the HCW participants’ qualitative answers. Finally, a priori coding, in which the codes are developed before looking at the data as opposed to inductive/open-coding in which codes are developed while examining the data, may have limited the depth of themes identified, particularly as the coding schemas were heavily informed by studies that used quantitative methods (Charmaz 2006).
Discussion
Prematunge et al. (2013) found that, with a few exceptions, motivators and barriers to accepting influenza vaccination were similar for both sINFLU and pH1N1 vaccines among the HCW participating in their study. This suggests that further refining vaccination campaigns to address common misperceptions and emphasize benefits of vaccination would likely improve vaccine uptake during seasonal influenza campaigns and during pandemics. In particular, targeting vaccination campaigns to highlight the altruistic nature of being vaccinated would reinforce the belief held by many vaccinated HCWs that influenza is a potentially serious disease for them personally and for those around them. Emphasizing that influenza vaccination is important in protecting and preventing infection of loved ones, patients, colleagues, and communities at large, and that non-vaccination has potentially negative effects on the surrounding community, would increase the effectiveness of vaccine promotion. Furthermore, Prematunge (2013) suggests that vaccination campaigns can be used to appeal to HCWs’ sense of professional obligation by reminding them that, due to the nature of their job, they have increased personal risk of infection and increased risk for transmitting the virus to a particularly susceptible population (i.e. hospital-based patients). Other studies investigating HCWs’ attitudes to vaccination have found similar results (Hollmeyer, Hayden et al. 2009, Hidiroglu, Ay et al. 2010, Poland 2010, Thomas, Jefferson et al. 2010, Lam, Chambers et al. 2010a, Hollmeyer, Hayden et al. 2013). In particular, Prematunge (2013) identifies three interventions that would address the barriers identified and potentially significantly improve vaccination rates of HCWs, including: 1) creating a positive, pro-vaccination workplace culture, where peers and supervisors encourage vaccination; 2) making it as easy as possible to become vaccinated, such as decreasing wait times for HCWs and increasing accessibility; and 3) ensuring consistent and clear communication strategies to address common fears and misconceptions.
A Pro-Vaccination Workplace Culture
Prematunge (2013) is one of the few studies that report on the influence of workplace policies and colleagues’ attitudes and behaviors on pH1N1 vaccination. This is consistent with a qualitative study (Yassi, Lockhart et al. 2010) on HCW perceptions of non-pandemic influenza policies. Using focus groups, Yassi, Lockhart et al. targeted the regions with the highest and lowest rates of HCW vaccination in British Columbia, where unvaccinated HCWs can be excluded from work without pay during an influenza outbreak (Yassi, Lockhart et al. 2010). Based on the four themes that emerged in their study (knowledge, communication, perceived punitive nature of workplace policy, and safety climate), the researchers reported the importance of using a comprehensive approach to promote a healthy workplace, integrating vaccination with other initiatives and not basing HCW vaccination on coercion (Yassi, Lockhart et al. 2010). Positive reinforcement from those in leadership positions has also been found to be important (Bish, Yardley et al. 2011). For example, a study in Turkey found that the Prime Minister’s public refusal to be vaccinated for pH1N1 had a significant negative impact on HCW willingness to be vaccinated (Savas and Tanriverdi 2010). Education campaigns alone have been found to be ineffective at improving HCW vaccination rates (Lam 2010b), highlighting the importance of encouraging a pro-vaccination culture in the workplace through holistic, long-term intervention strategies such as respectful policies, strong leadership, and encouragement from peers.
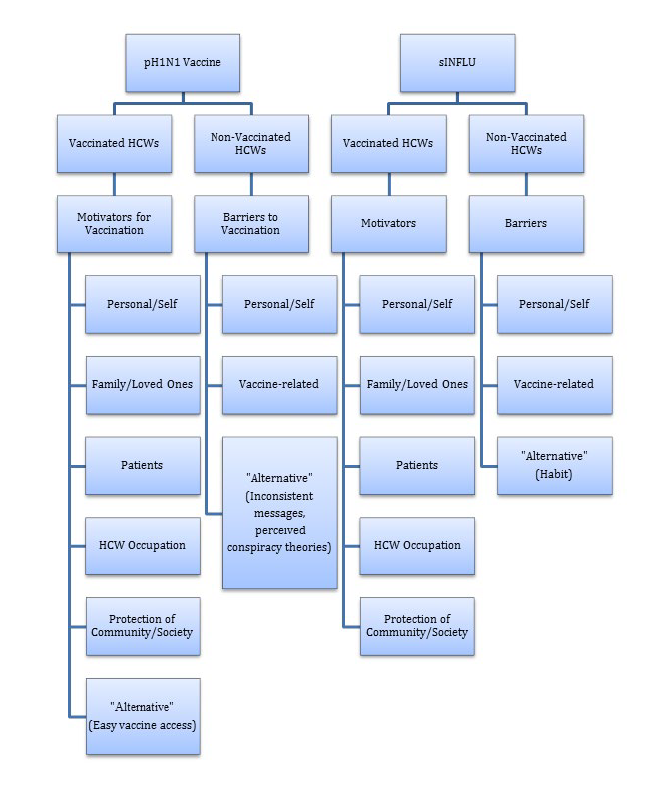
Personal / Self
- Personal health
- Personal safety and protection
- Reduced infection risk to self
- Fear of infection *
- The pandemic nature of pH1N1 *
- Perceptions that pH1N1 is more severe than sINFLU *
Family / Loved Ones
- Safety and protection of loved ones
- Preventing transmission to loved ones
- Acknowledged increased risk among loved ones (i.e. young children, elderly relatives, chronic health conditions)
Patients
- Patient safety and protection
- Recognizing their own role in preventing influenza transmission to patients at point of work/care
HCW Occupation
- Potential increases in workload (from influenza-related absenteeism)
- Professional obligation
- Occupational hazard of the job
- Work place requirement
- Encouragement from colleagues and supervisors
Community
- Limiting general influenza spread in others
Alternative
- Convenient access to pH1N1 vaccine *
- Free access to the vaccine *
Figure 2: Further description of each motivator as described by vaccinated HCWs for both pH1N1 and sINFLU vaccination. Items marked with an asterisk (*) refer to pH1N1 vaccination only. No items were specific to sINFLU vaccination.
Personal
- Personal immune function is sufficient and can handle an influenza infection so vaccination is not necessary
- Do not get the flu
- They are never sick
- Perceive themselves to be at low risk for infection
- “Do not believe in immunization”
- Personal choice
- Previously experienced side-effects to an influenza vaccine, such as a sore arm, headache, or feeling sick
- Promote one’s “natural immunity” through exposure to the virus without concurrent immunization **
Vaccine-related
- Vaccine safety, lack of trust of the vaccine
- Vaccine ingredients, issues such as adverse-effects, allergies, exposure to chemicals (e.g. thimerosal, mercury, and adjuvants) used in manufacturing
- Vaccine-induced adverse reactions experienced by others (e.g. HCW friends, family members)
- Lack of vaccine effectiveness
- Belief of vaccination causing influenza or influenza-like illness
- Perceptions that the pH1N1 vaccine was novel and different from sINFLU, rushed into development, and not as vigorously assessed for vaccine safety issues or long-term side effects (compared to conventional sINFLU vaccines) *
- Inability to prevent influenza because of inaccuracy of predicting seasonal flu strains **
Alternative
- Lack of clear and consistent communication about the vaccine *
- Perception that pH1N1 was exaggerated by various groups (e.g. media, pharmaceutical companies, government organizations, and other public health authorities) for conspiracy related reasons and/or monetary gain *
- Do not receive sINFLU vaccination out of habit **
Figure 3: Further description of each barrier to accepting vaccination reported by unvaccinated HCWs for both pH1N1 and sINFLU vaccination. Items marked with a single asterisk (*) are specific to pH1N1 vaccination, and items marked with a double asterisk (**) are specific to sINFLU vaccination.
Easy Vaccine Access
Convenience in accessing vaccination is well documented to be a significant factor positively influencing vaccination rates of HCWs (Music 2012, Pianosi, Chobotuk et al. 2012). Prematunge (2013) notes that initiatives taken by TOH during pH1N1 to increase ease of accessing pH1N1 vaccination were successful in eliminating this as a barrier to pH1N1 vaccination and so suggests that these interventions also be used with sINFLU vaccination programs. Successful interventions implemented at TOH during pH1N1 that led to improved HCW vaccination rates included extended vaccine clinics, weekend vaccine clinics that accommodated staff and staff family members, peer-to-peer vaccination programs, and notices from hospital administration to departmental supervisors requesting that they promote vaccination. Prematunge (2013) cites several studies (Poland, Tosh et al. 2005, Hollmeyer, Hayden et al. 2009, Lam, Chambers et al. 2010a) conducted prior to pH1N1 that found inconvenient vaccine access, scheduling issues, and lack of time to be major barriers in becoming vaccinated for sINFLU, and similar results have been found specific to other pH1N1 HCW vaccination campaigns in Canada (Kraut, Graff et al. 2011, Long 2013) and globally (DiBiase, Davis et al. 2011, Al-Tawfiq 2012, Rebmann, Iqbal et al. 2012).
Clear communication
Barriers to influenza vaccination were found to be mostly similar between pH1N1 and sINFLU by Prematunge (2013), with a large proportion of unvaccinated HCWs reporting a lack of confidence in influenza vaccines. Specifically regarding pH1N1, non-vaccinated HCWs in Prematunge’s study (2013) cited the following barriers to vaccination: limited research on the vaccine, receiving conflicting messages about the vaccine, and compromised vaccine safety due to the accelerated vaccine development processes. Although there are no other qualitative studies specific to Canadian HCWs, a qualitative study in Turkey used focus groups to investigate primary healthcare workers’ perceptions of pH1N1 vaccination and found willingness to become vaccinated depended on their source of information (Hidiroglu, Ay et al. 2010). HCWs who depended on the media for information were more likely to refuse pH1N1 vaccination, believing that even though they were susceptible to infection, their strong immune systems would be adequate protection. This group also failed to recognize their role in potential transmission to others. In contrast, HCWs that depended on evidence-based information from health authorities, professional organizations, and the World Health Organization were more likely to accept vaccination (Hidiroglu, Ay et al. 2010). These findings were supported by quantitative studies in Turkey (Savas and Tanriverdi 2010) and Canada (Kaboli, Astrakianakis et al. 2010) that found low vaccination of HCWs related to perceptions of limited effectiveness and questionable safety of the vaccine, prompting these researchers to call for improved communication from media and government officials. Indeed, analyses of pH1N1 media coverage found that vaccination campaigns often based reporting on weak science and failed to mention vaccine safety (Laing 2011, Rachul, Ries et al. 2011). Other studies and reviews have also found that clear communication that alleviates fears regarding pH1N1 and sINFLU vaccine safety are required to improve HCW vaccination rates (Gallant, Vollman et al. 2008, Poland 2010, Blasi, Aliberti et al. 2012, Music 2012, Quach, Pereira et al. 2013b).
Effective communication must speak to HCWs’ apprehensions and improve their confidence in vaccine safety and effectiveness to improve HCW vaccination rates. These findings also highlight the critical need for clear, consistent messaging to prevent the propagation of inaccurate information, especially important among HCWs who may be educating or influencing other non-HCWs (Dube, Fannie et al. 2011). This is particularly relevant with respect to the alarming finding that several sINFLU non-vaccinated HCWs mistakenly believe that not getting vaccinated will “enhance natural immunity,” for which there is no scientifically valid support (Prematunge 2013). This prompts Prematunge (2013) to call for HCWs to have increased access to information about influenza transmission, risk and clinical signs of vaccine-related side-effects, and vaccine ingredients and the development process. She suggests that this may ease HCW concerns about vaccination and bridge knowledge gaps. Indeed, other studies have found that HCWs are more likely to accept vaccination if they understand influenza vaccination risks and general immune function (Kaboli, Astrakianakis et al. 2010, Lam, Chambers et al. 2010a, Hakim, Gaur et al. 2011, Mitchell, Ogunremi et al. 2012, Prematunge, Corace et al. 2012).
Transparency in communication is thought to be especially critical in pandemic situations, when it is more likely that pandemic vaccine processes may be perceived as inferior to sINFLU vaccine practices and standards. Specific to pH1N1, Prematunge (2013) found that HCWs’ refusal to be vaccinated was influenced by their negative perceptions of pandemic management by different nonclinical groups, such as the media, pharmaceutical companies, government organizations, and public health authorities. Issues stated such as lack of transparency in the decision-making process by authorities and a lack of trust in the competency of the decision-makers have also been reported in other studies (Nhan, Laprise et al. 2012, Rosella, Wilson et al. 2013). Issues of perceived monetary gain and conspiracies as barriers to vaccination during the pandemic have also been reported by other studies, including a qualitative study investigating the Canadian public’s perceptions of pH1N1 vaccination based on online comments (Henrich and Holmes 2011). Although not addressing perceptions of vaccination specifically, a qualitative study in Manitoba exploring public health nurses’ pH1N1 experiences noted that inconsistent and delayed messages from health authorities also sometimes conflicted with media reports, contributing to the confusion and mistrust around pH1N1 vaccination recommendations (Long 2013). In contrast, surveys of Canadian pediatricians’ knowledge, attitudes, and practices regarding pH1N1 and its prevention by vaccination before and soon after the launch of the pH1N1 vaccination campaign, showed an improvement in the pediatricians’ confidence in pH1N1 vaccine safety and their willingness to recommend vaccination to their patients. This may, at least partly, be due to information disseminated by public health authorities contributing toward an effective campaign to educate physicians on the pH1N1 vaccine (Dube, Fannie et al. 2011).
Future Work
Premtunge (2013) calls for further qualitative studies to better understand factors influencing the perceptions that HCWs appeared to have regarding pH1N1, such as perceiving pH1N1 to be more serious than sINFLU and being fearful of pH1N1, that lead to increased vaccination rates compared to sINFLU. In particular, it is important to understand the “emotional epidemiology” of diverse reactions to a novel vaccine during a pandemic. One physician in New York City described a shift in demand for a vaccine from very high, when pH1N1 was just emerging and panic was high, to indifference or suspicion once a vaccine was available, when the pandemic was later perceived to have been exaggerated (Ofri 2009). In addition, better understanding of vaccine perceptions could be achieved by improving healthcare facilities’ ability to track HCW immunization status, as noted by two Canadian studies, one using quantitative methods (Quach, Pereira et al. 2013c) and one using qualitative methods (Quach, Pereira et al. 2013a).
Limitations
Although an extensive search was conducted for relevant studies, it is possible that one or more studies may have been overlooked as the subject concerns multiple disciplines and studies may appear in various publications and/or databases. Non-English studies may also exist.
Conclusion
Many of the motivators and barriers associated with accepting vaccination were found by Prematunge (2013) to be similar for seasonal flu and pandemic H1N1 vaccination campaigns, suggesting that robust annual seasonal flu vaccination programs will also contribute to effective vaccination campaigns in pandemic situations. Creating a pro-vaccination culture in the workplace, improving vaccine access, and ensuring transparency and consistency in communications will have secondary effects during the next pandemic and improve HCW vaccination rates. In particular, targeted vaccination campaigns to alleviate vaccine-related misconceptions and address knowledge gaps are critical in ensuring HCWs are confident in the benefits of mass immunization campaigns. Additionally, to improve HCWs’ acceptance of vaccination during public health emergencies, it is important to address motivators and barriers that were specific to pH1N1, such as convenience in getting vaccinated and perceptions of trust in vaccine manufacturing and distributing processes. Further qualitative research will allow better command of the underlying factors influencing HCWs’ perceptions of seasonal and pandemic-specific influenza vaccination, leading to increased insight into HCWs’ motivations and barriers to accepting vaccination. Through understanding HCWs’ behaviors during a novel disease outbreak, Canada’s healthcare system can be better prepared for the next pandemic.
References
Al-Tawfiq, J. A. (2012). Willingness of health care workers of various nationalities to accept H1N1 (2009) pandemic influenza A vaccination. Ann Saudi Med 32(1): 64-67.
Bish, A., L. Yardley, A. Nicoll and S. Michie (2011). Factors associated with uptake of vaccination against pandemic influenza: a systematic review. Vaccine 29(38): 6472-6484.
Blasi, F., S. Aliberti, M. Mantero and S. Centanni (2012). Compliance with anti-H1N1 vaccine among healthcare workers and general population. Clin Microbiol Infect 18 Suppl 5: 37-41.
Brewer, N. T., G. B. Chapman, F. X. Gibbons, M. Gerrard, K. D. McCaul and N. D. Weinstein (2007). Meta-analysis of the relationship between risk perception and health behavior: the example of vaccination. Health Psychol 26(2): 136-145.
Brien, S., J. C. Kwong and D. L. Buckeridge (2012). The determinants of 2009 pandemic A/H1N1 influenza vaccination: a systematic review. Vaccine 30(7): 1255- 1264.
Bryce, E., J. Embree, G. Evans, L. Johnston, K. Katz, A. McGeer, D. Moore, V. Roth, A. Simor, K. Suh and M. Vearncombe (2012). AMMI Canada position paper: 2012 Mandatory influenza immunization of health care workers. Can J Infect Dis Med Microbiol 23(4): e93-95.
Burls, A., R. Jordan, P. Barton, B. Olowokure, B. Wake, E. Albon and J. Hawker (2006). Vaccinating healthcare workers against influenza to protect the vulnerable—is it a good use of healthcare resources?: a systematic review of the evidence and an economic evaluation. Vaccine 24(19): 4212-4221.
CDC (2009). Use of influenza A (H1N1) 2009 monovalent vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009. MMWR Recomm Rep 58(RR-10): 1-8.
Charania, N. A. and L. J. S. Tsuji (2011). The 2009 H1N1 pandemic response in remote First Nation communities of Subarctic Ontario: barriers and improvements from a health care services perspective. International Journal of Circumpolar Health 70(5): 564-575.
Charmaz, K. (2006). Constructing Grounded Theory: A practical guide through qualitative analysis, SAGE Publications, Inc.
Corace, K., C. Prematunge, A. McCarthy, R. C. Nair, V. Roth, T. Hayes, K. N. Suh, L. Balfour and G. Garber (2013). Predicting influenza vaccination uptake among health care workers: what are the key motivators? Am J Infect Control 41(8): 679-684.
DiBiase, L. M., S. E. Davis, R. Rosselli and J. Horney (2011). Evaluation of the implementation of the H1N1 pandemic influenza vaccine in local health departments (LHDs) in North Carolina. Vaccine 29(23): 3969-3976.
Dube, E., D. Fannie, G. Vladimir, B. A. Julie, S. Chantal, L. France, B. D. Francois, M. Shelly, G. Ian and B. Nicole (2011). A(H1N1) pandemic influenza and its prevention by vaccination: paediatricians’ opinions before and after the beginning of the vaccination campaign. BMC Public Health 11: 128.
Embree, J. (2010). Pandemic 2009 (A) H1N1 influenza (swine flu)—the Manitoba experience. Biochem Cell Biol 88(4): 589-593.
Gallant, D., A. Vollman and S. Sethi (2008). Influenza vaccination by registered nurses: a personal decision. Can J Infect Contr Rev Canad Prev Infect. 24(1): 18- 22, 24, 26.
Hakim, H., A. H. Gaur and J. A. McCullers (2011). Motivating factors for high rates of influenza vaccination among healthcare workers. Vaccine 29(35): 5963-5969.
Health, O. C. M. O. o. and A. King (2010). The H1N1 Pandemic: How Ontario Fared: a Report, Ministry of Health and Long-Term Care.
Henrich, N. and B. Holmes (2011). What the public was saying about the H1N1 vaccine: perceptions and issues discussed in on-line comments during the 2009 H1N1 pandemic. PLoS One 6(4): e18479.
Herzog, R., M. J. Alvarez-Pasquin, C. Diaz, J. L. Del Barrio, J. M. Estrada and A. Gil (2013). Are healthcare workers’ intentions to vaccinate related to their knowledge, beliefs and attitudes? A systematic review. BMC Public Health 13: 154.
Hidiroglu, S., P. Ay, A. Topuzoglu, C. Kalafat and M. Karavus (2010). Resistance to vaccination: the attitudes and practices of primary healthcare workers confronting the H1N1 pandemic. Vaccine 28(51): 8120-8124.
Hidiroglu, S., P. Ay, A. Topuzoglu, C. Kalafat and M. Karavus (2010). Resistance to vaccination: The attitudes and practices of primary healthcare workers confronting the H1N1 pandemic. Vaccine 28(51): 8120-8124.
Hofmann, F., C. Ferracin, G. Marsh and R. Dumas (2006). Influenza vaccination of healthcare workers: a literature review of attitudes and beliefs. Infection 34(3): 142-147.
Hollmeyer, H., F. Hayden, A. Mounts and U. Buchholz (2013). Review: interventions to increase influenza vaccination among healthcare workers in hospitals. Influenza Other Respir Viruses 7(4): 604-621.
Hollmeyer, H. G., F. Hayden, G. Poland and U. Buchholz (2009). Influenza vaccination of health care workers in hospitals—a review of studies on attitudes and predictors. Vaccine 27(30): 3935-3944.
Kaboli, F., G. Astrakianakis, G. Li, J. Guzman, T. Donovan and M. Naus (2010). Influenza vaccination and intention to receive the pandemic H1N1 influenza vaccine among healthcare workers of British Columbia, Canada: a cross-sectional study. Infect Control Hosp Epidemiol 31(10): 1017-1024.
Kendal, A. P. and N. E. MacDonald (2010). Influenza pandemic planning and performance in Canada, 2009. Can J Public Health 101(6): 447-453.
Khandaker, G., A. Dierig, H. Rashid, C. King, L. Heron and R. Booy (2011). Systematic review of clinical and epidemiological features of the pandemic influenza A (H1N1) 2009. Influenza Other Respi Viruses 5(3): 148-156.
Kraut, A., L. Graff and D. McLean (2011). Behavioral change with influenza vaccination: factors influencing increased uptake of the pandemic H1N1 versus seasonal influenza vaccine in health care personnel. Vaccine 29(46): 8357-8363.
Krippendorff, K. (2012). Content analysis: An introduction to its methodology, Sage.
Kumar, A., R. Zarychanski, R. Pinto, D. J. Cook, J. Marshall, J. Lacroix, T. Stelfox, S. Bagshaw, K. Choong, F. Lamontagne, A. F. Turgeon, S. Lapinsky, S. P. Ahern, O. Smith, F. Siddiqui, P. Jouvet, K. Khwaja,
L. McIntyre, K. Menon, J. Hutchison, D. Hornstein, A. Joffe, F. Lauzier, J. Singh, T. Karachi, K. Wiebe, K. Olafson, C. Ramsey, S. Sharma, P. Dodek, M. Meade, R. Hall and R. A. Fowler (2009). Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA 302(17): 1872-1879.
Laing, A. (2011). The H1N1 crisis: Roles played by government communicators, the public and the media. Journal of Professional Communication 1(1): 11.
Lam, P.-P. (2010b). Approaches to implementing an influenza vaccine decision aid for healthcare personnel. MR69024 M.Sc., University of Ottawa (Canada).
Lam, P. P., L. W. Chambers, D. M. MacDougall and A. E. McCarthy (2010a). Seasonal influenza vaccination campaigns for health care personnel: systematic review. CMAJ 182(12): E542-548.
Long, M. M. (2013). Response to the 2009 H1N1 influenza pandemic: Manitoba public health nurses’ experience. FGS – Electronic Theses & Dissertations (Public)
Manitoba Heritage Theses.
Looijmans-van den Akker, I., J. J. van Delden, T. J. Verheij, G. A. van Essen, M. A. van der Sande, M. E. Hulscher and E. Hak (2009). Which determinants should be targeted to increase influenza vaccination uptake among health care workers in nursing homes? Vaccine 27(34): 4724- 4730.
Marsh, E. E. and M. D. White (2006). Content analysis: A flexible methodology. Library trends 55(1): 22-45.
Mitchell, R., T. Ogunremi, G. Astrakianakis, E. Bryce, R. Gervais, D. Gravel, L. Johnston, S. Leduc, V. Roth, G. Taylor, M. Vearncombe and C. Weir (2012). Impact of the 2009 influenza A (H1N1) pandemic on Canadian health care workers: a survey on vaccination, illness, absenteeism, and personal protective equipment. Am J Infect Control 40(7): 611-616.
Mok, E., S. H. Yeung and M. F. Chan (2006). Prevalence of influenza vaccination and correlates of intention to be vaccinated among Hong Kong Chinese. Public Health Nurs 23(6): 506-515.
Monto, A. S. (2010). Seasonal influenza and vaccination coverage. Vaccine 28: D33-D44.
Music, T. (2012). Protecting patients, protecting healthcare workers: a review of the role of influenza vaccination. Int Nurs Rev 59(2): 161-167.
Neuendorf, K. (2002). The content analysis guidebook. 2nd ed. Thousand Oakes, California, SAGE Publications.
Nhan, C., R. Laprise, M. Douville-Fradet, M. E. Macdonald and C. Quach (2012). Coordination and resource-related difficulties encountered by Quebec’s public health specialists and infectious diseases/medical microbiologists in the management of A (H1N1) – a mixed-method, exploratory survey. BMC Public Health 12: 115.
Nichol, K. L. and J. J. Treanor (2006). Vaccines for seasonal and pandemic influenza. Journal of Infectious Diseases 194(Supplement 2): S111-S118.
Ofri, D. (2009). The Emotional Epidemiology of H1N1 Influenza Vaccination. New England Journal of Medicine 361(27): 2594-2595.
Orr, P. (2000). Influenza vaccination for health care workers: A duty of care. The Canadian journal of infectious diseases 11(5): 225-226.
Padgett, D. (2012). Qualitative and Mixed Methods in Public Health, SAGE Publications, Inc.
Pianosi, K., T. Chobotuk, B. Halperin and S. Halperin (2012). Influenza immunization practices and policies for health care students in Canada. CJIDMM 24(4): 195-201.
Poland, G. A. (2010). The 2009-2010 influenza pandemic: effects on pandemic and seasonal vaccine uptake and lessons learned for seasonal vaccination campaigns. Vaccine 28 Suppl 4: D3-13.
Poland, G. A., C. L. Ofstead, S. J. Tucker and T. J. Beebe (2008). Receptivity to mandatory influenza vaccination policies for healthcare workers among registered nurses working on inpatient units. Infect Control Hosp Epidemiol 29(2): 170-173.
Poland, G. A., P. Tosh and R. M. Jacobson (2005). Requiring influenza vaccination for health care workers: seven truths we must accept. Vaccine 23(17): 2251-2255.
Prematunge, C. (2013). An Analysis of Healthcare Worker Attitudes & Barriers to Influenza Vaccination. MR87213 M.Sc., University of Ottawa (Canada).
Prematunge, C., K. Corace, A. McCarthy, R. C. Nair, R. Pugsley and G. Garber (2012). Factors influencing pandemic influenza vaccination of healthcare workers – a systematic review. Vaccine 30(32): 4733-4743.
Quach, S., J. A. Pereira, J. S. Hamid, L. Crowe, C. L. Heidebrecht, J. C. Kwong, M. Guay, N. S. Crowcroft, A. McGeer and L. W. Chambers (2013c). Measuring influenza immunization coverage among health care workers in acute care hospitals and continuing care organizations in Canada. American journal of infection control 41(4): 340-344.
Quach, S., J. A. Pereira, C. L. Heidebrecht, J. C. Kwong, M. Guay, L. Crowe, S. Quan and J. A. Bettinger (2013a). Health care worker influenza immunization rates: The missing pieces of the puzzle. American journal of infection control 41(8): 685-690.
Quach, S., J. A. Pereira, J. C. Kwong, S. Quan, L. Crowe, M. Guay and J. A. Bettinger (2013b).Immunizing health care workers against influenza: A glimpse into the challenges with voluntary programs and considerations for mandatory policies. American journal of infection control 41(11): 1017-1023.
Rachul, C. M., N. M. Ries and T. Caulfield (2011). Canadian newspaper coverage of the A/H1N1 vaccine program. Can J Public Health 102(3): 200-203.
Rebmann, T., A. Iqbal, J. Anthony, R. C. Knaup, K. S. Wright and E. B. Peters (2012). H1N1 influenza vaccine compliance among hospital- and non-hospital-based healthcare personnel. Infect Control Hosp Epidemiol 33(7): 737-744.
Rosella, L. C., K. Wilson, N. S. Crowcroft, A. Chu, R. Upshur, D. Willison, S. L. Deeks, B. Schwartz, J. Tustin, D. Sider and V. Goel (2013). Pandemic H1N1 in Canada and the use of evidence in developing public health policies—a policy analysis. Soc Sci Med 83: 1-9.
Savas, E. and D. Tanriverdi (2010). Knowledge, attitudes and anxiety towards influenza A/H1N1 vaccination of health-care workers in Turkey. BMC Infect Dis 10: 281.
Saxen, H. and M. Virtanen (1999). Randomized, placebo-controlled double blind study on the efficacy of influenza immunization on absenteeism of health care workers. The Pediatric infectious disease journal 18(9): 779-783.
Scott, K. (2010). Pandemic influenza H1N1 2009—the Canadian experience. J Popul Ther Clin Pharmacol 17(3): e358- 362.
Spika, J. S. and D. Butler-Jones (2009). Pandemic influenza (H1N1): our Canadian response. Can J Public Health 100(5): 337-339.
Standards, C. (2010). Voices from the H1N1 Influenza Pandemic Front Lines: A White Paper About How Canada Could Do Better Next Time. CSA Standards Roundtable on Healthcare and Emergency Service Sector Pandemic Preparedness. D. Doug Morton, Health and Safety CSA Standards; Dr. Allan Holmes, moderator.
Thomas, R. E., T. Jefferson and T. J. Lasserson (2010). Influenza vaccination for healthcare workers who work with the elderly. Cochrane Database Syst Rev(2): CD005187.
Weinstein, R. A., C. B. Bridges, M. J. Kuehnert and C. B. Hall (2003). Transmission of influenza: implications for control in health care settings. Clinical Infectious Diseases 37(8): 1094-1101.
Wynn, A. and K. M. Moore (2012). Integration of primary health care and public health during a public health emergency. Am J Public Health 102(11): e9-e12.
Yassi, A., K. Lockhart, J. A. Buxton and I. McDonald (2010). Vaccination of health care workers for influenza: promote safety culture, not coercion. Can J Public Health 101(7): S41-S45.
Zarocostas, J. (2009). Healthcare workers should get top priority for vaccination against A/H1N1 flu, WHO says. BMJ 339.
